Sponsors/CRO
Discover how our solutions for Sponsors and CROs can
help you meet your goals in research today and tomorrow.
At BV Medical Research
As a full-service phase II-IV clinical trial Site Management Organisation, we serve a variety of human health industries to provide the highest standards in Clinical Trials.
Our network is fully equipped for decentralized trials (DCT) as we strive to accelerate research through innovation. BV Medical Research understands clinical issues that drive research: our services ensure patient-centered protocols are properly executed and top-quality data is collected without sacrificing regulatory compliance.

Our Commitment To Unrivalled Data Integrity
BV Medical Research utilizes active clinical trial quality assurances that detail proactive procedures ensuring all trials are compliant with Health Canada, ICH and GCP guidelines, 21 CFR/FDA regulations, sponsor requirements, protocol requirements and BV Medical Research policies.
Our quality assurance plan includes comprehensive investigative protocol framework and documentation of site work processes to ensure procedures are followed.
Our investigators are highly committed to the oversight of each patient's data and to ensuring adherence to all applicable regulatory guidelines including PHIPA, HIPAA, and ISO 27001 compliancy.
Our industry-leading technology allows for valid, auditable and accurate data. Cloud capabilities allow for rapid and seamless accessibility of data throughout our network. You’ll find it’s easier to work with us when you have on-demand access to high-quality data and remote collaboration capabilities.
Experienced Site Management
Our sites are dedicated clinical trial facilities featuring parking for trial participants, clinical monitor rooms, highly secured supplies rooms for investigative products, and secure storage for study material.
Additional site resources include:
Dedication. Quality. Results.
BV Medical Research is a dedicated team comprised of physicians and other clinical research professionals who are passionate about patient care and delivering quality clinical trial data through state of the art technology.
We use high-fidelity database computing technology, allowing for less expensive and more efficient processing of critical data. Our team is always open to learning and feedback, and committed to make the necessary changes to exceed standards.

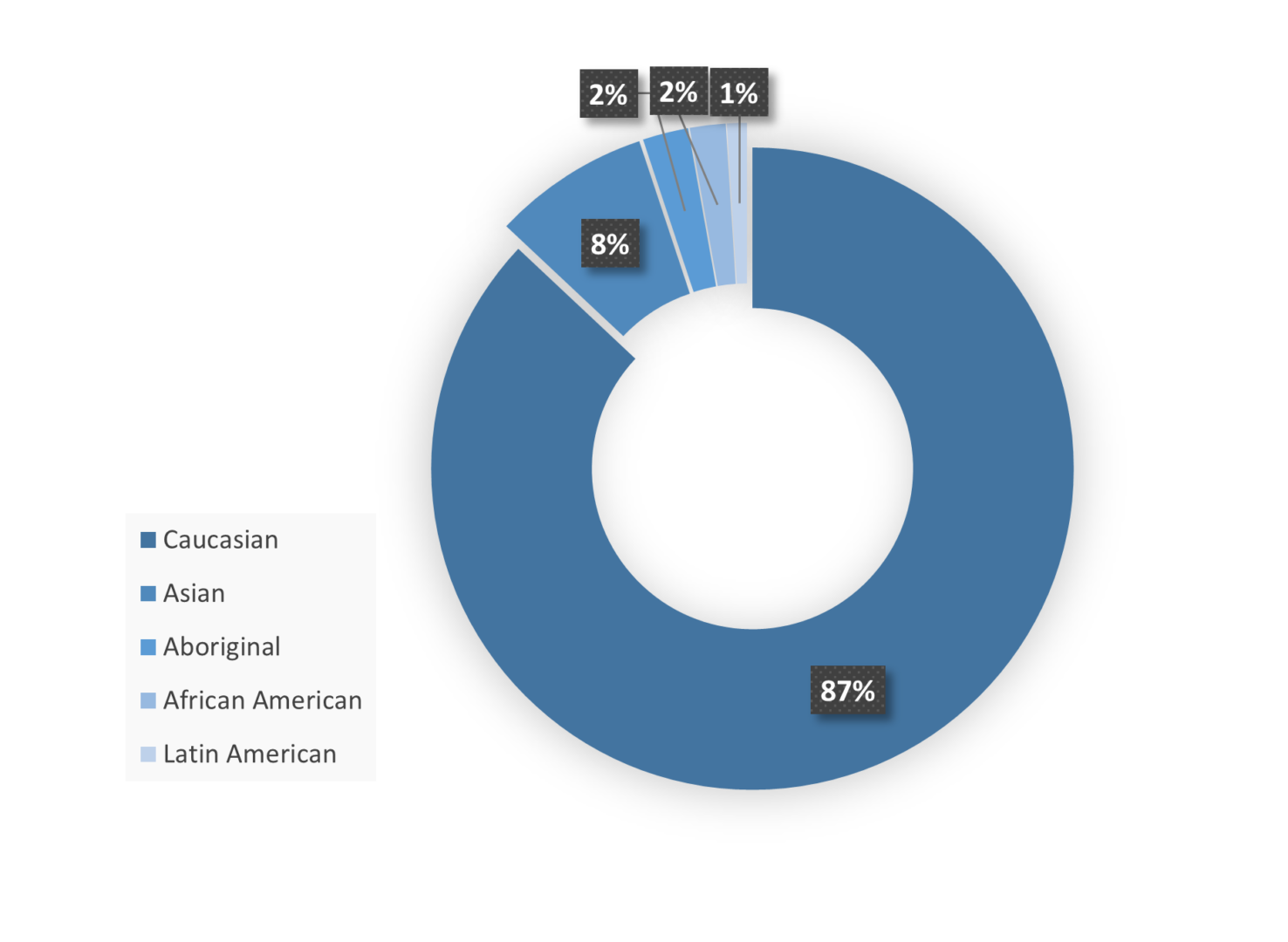
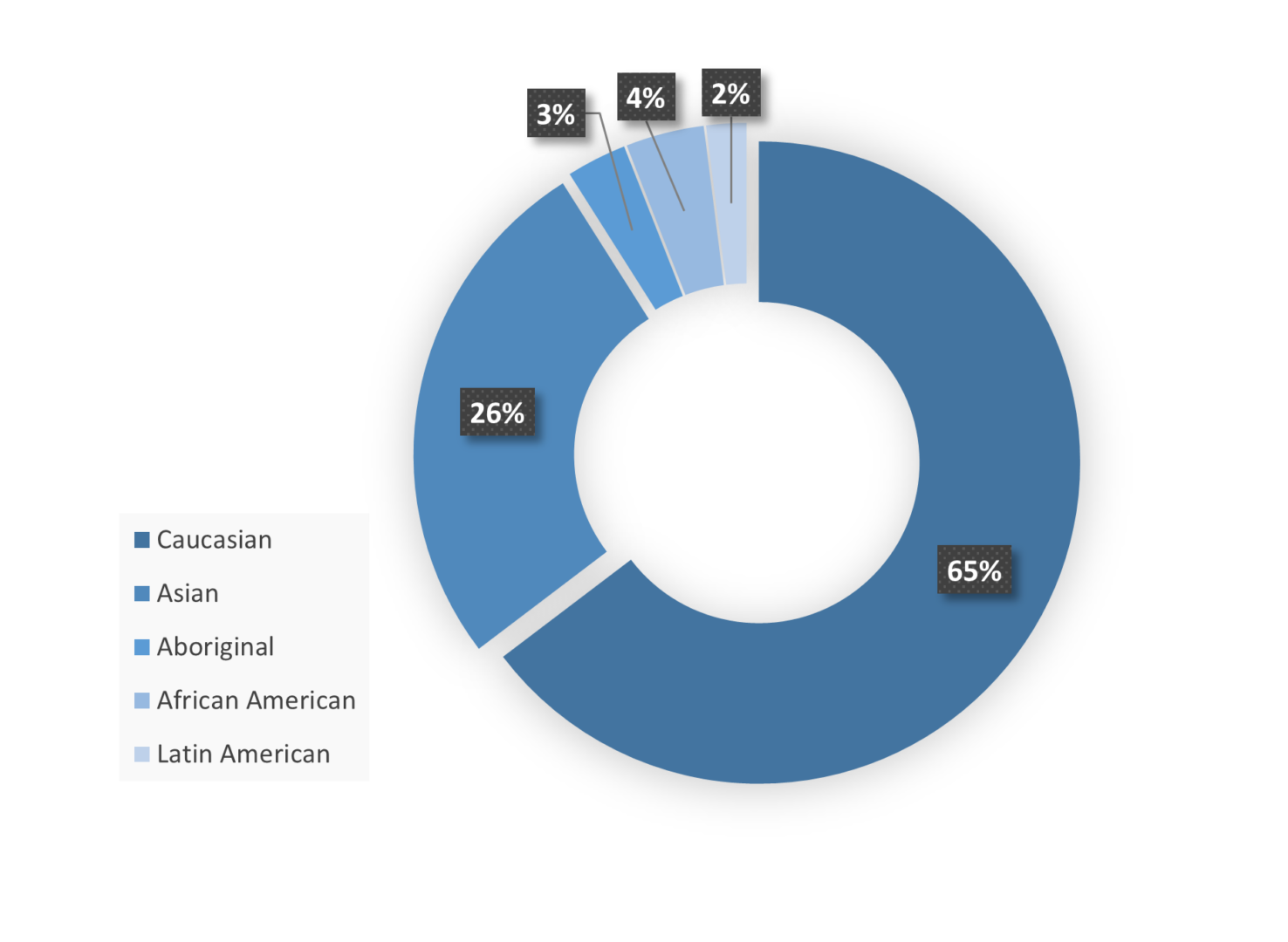
Site Regional Demographics
Therapeutic Expertise
Internal Medicine
Women's Health
Immunology
Allergy
Dermatology
Cardiology
Family Medicine
Virology (HIV)
Pediatrics
Vaccines
Respirology
Endocrine and Metabolics